Contents
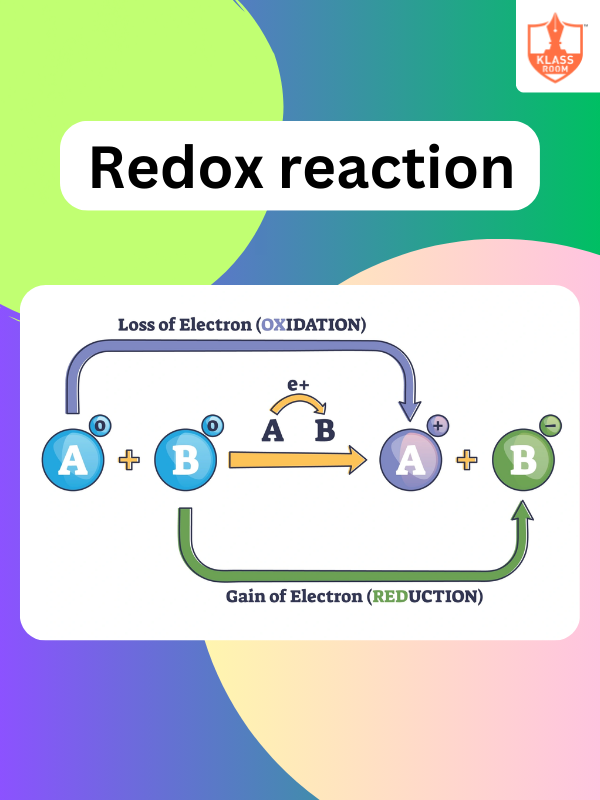
Redox Reaction
Description: Redox Reaction involves the transfer of electrons between species, resulting in oxidation and reduction processes.

Description: Redox Reaction involves the transfer of electrons between species, resulting in oxidation and reduction processes.
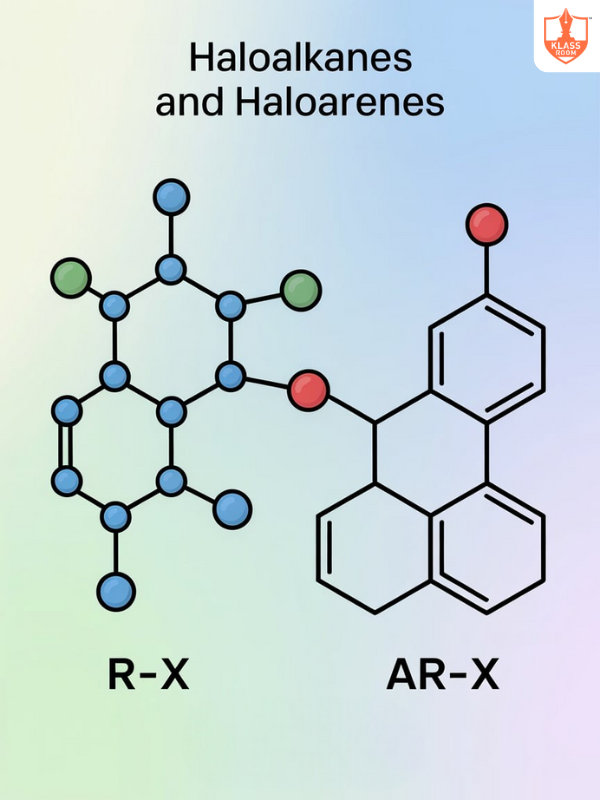

Board: State Board
Stream: Science
Standard: XI
Course: Chemistry
Know More.png)



Board: State Board
Stream: Science
Standard: XI
Course: Chemistry
Know More
Board: State Board
Stream: Science
Standard: XI
Course: Chemistry
Know More
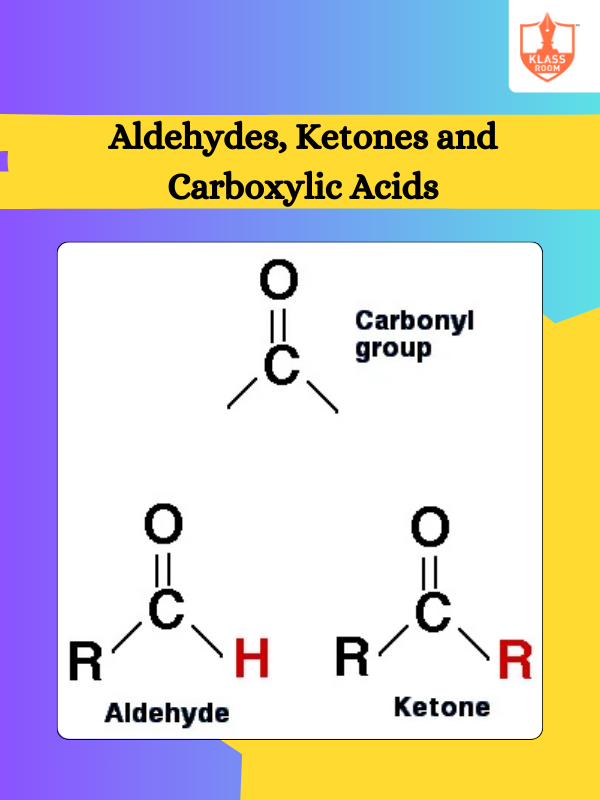
Board: State Board
Stream: Science
Standard: XII
Course: Chemistry
Know More
Board: State Board
Stream: Science
Standard: XII
Course: Chemistry
Know More
Board: State Board
Stream: Science
Standard: XII
Course: Chemistry
Know More