Contents

Surface Chemistry - I
Description: Surface Chemistry - I explores adsorption, surface phenomena, catalysts, and the interactions at solid-liquid interfaces.

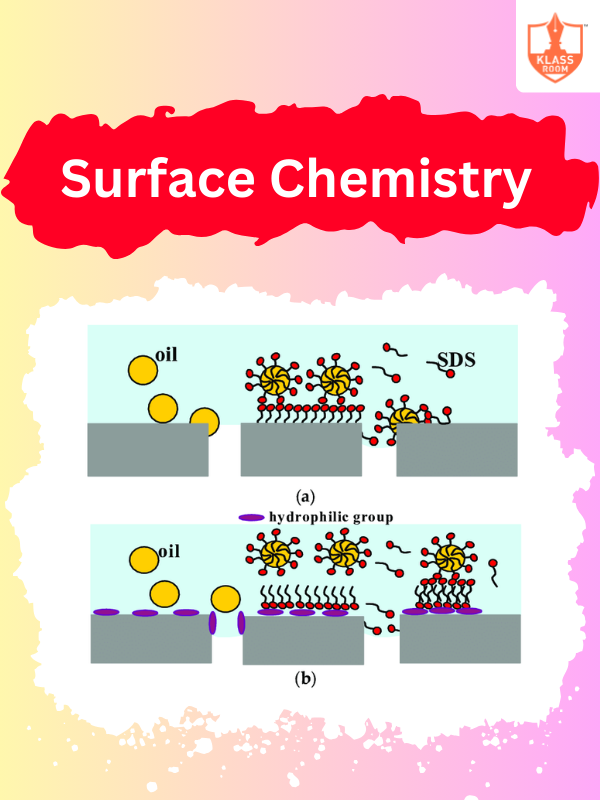
Surface Chemistry - II
Description: Surface Chemistry - II focuses on colloids, emulsions, foams, and the stability of dispersed systems.


.png)